Schöpfstr. 41
6020 Innsbruck
Fax: +43 512 9003 70800
Email: michaela.kress@i-med.ac.at
Website: https://www.i-med.ac.at/dpmp/physiologie/
Research year
Research Branch (ÖSTAT Classification)
301407, 106006
Keywords
auditory physiology, epithelial physiology, iPSC derived human model systems, nociception, non-coding RNA, and Voltage-gated calcium channels
Research Focus
Research at the Institute of Physiology focuses on basic and preclinical translational research on the (patho-)physiology of skeletal muscle, epithelial organs, and the nervous system and employs an integrated, interdisciplinary approach to explore physiological processes in cellular models, organs and recently model systems derived from human iPSCs (induced pluripotent stem cells). Particular innovation potential arises from ongoing projects on sensory functions, calcium channels, nuclear kinases, the gut microbiome and non-coding RNAs.
General Facts
The institute is heavily involved in medical research as well as in teaching human physiology to undergraduate as well as graduate students at all levels. Understanding healthy functions is essential for exploring the pathogenesis of common diseases, such as neurological disorders or chronic pain. The institute’s researchers engage in cutting-edge research on nociception, control of muscle contraction, auditory function, calcium signalling, cell membranes and epithelial physiology. We employ a wide range of models and techniques including cell culture, functional imaging, gene and protein expression, calcium microfluorimetry, high-resolution live microscopy and electrophysiology, and we team up with local, national and international consortia. We are grateful for the funding provided by the European Commission, FWF, FFG and others. The FWF-Docfunds project CavX-2 is co-chaired by M. Campiglio; M. Kress is a beneficiary of MSCA-ITN TOBeATPAIN funded by the European Commission.
Devices & Services
Two-Photon Microscopy (Core facility Biooptics)
Research
Neurobiology of Pain
Univ.-Prof. Dr. Michaela Kress
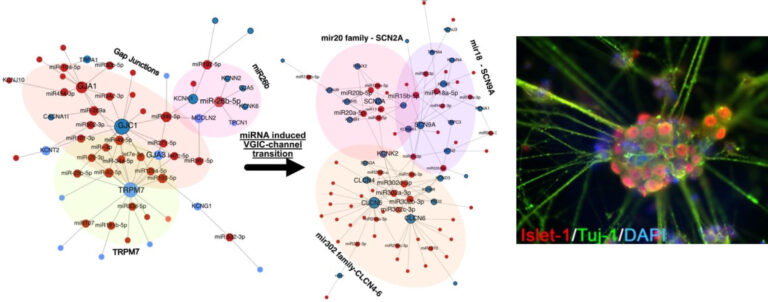
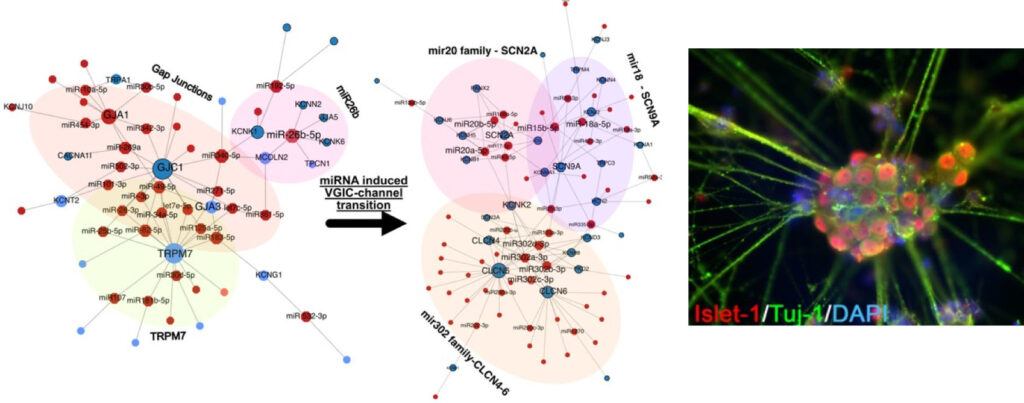
Chronic pain disorders affecting approximately 20% of people represent a major socioeconomic burden on society as well as patients and their families worldwide. The Kress lab explores deregulated molecular signalling along the pain pathway contributing to pain pathogenesis with a particularly focus on neuro-immune interactions involving cytokines, such as interleukin-6 and its signal transducer gp130, and mRNA::non-coding RNA interactions. In order to bridge the translational gap between preclinical research and the lack of efficient therapies for patients, iPSC derived models of nociception are optimised to approximate native human functionalities (Fig. 1) with a comprehensive methodological approach exploiting cutting-edge electrophysiology, molecular biology, complex bioinformatics and the NOCICEPTRA tool available for the scientific community.
Calcium Channel Function
Univ.-Prof. Dr. Bernhard E. Flucher
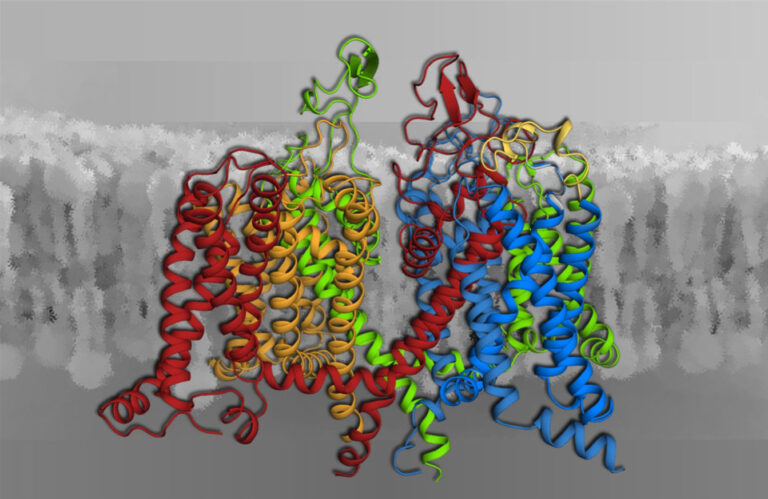
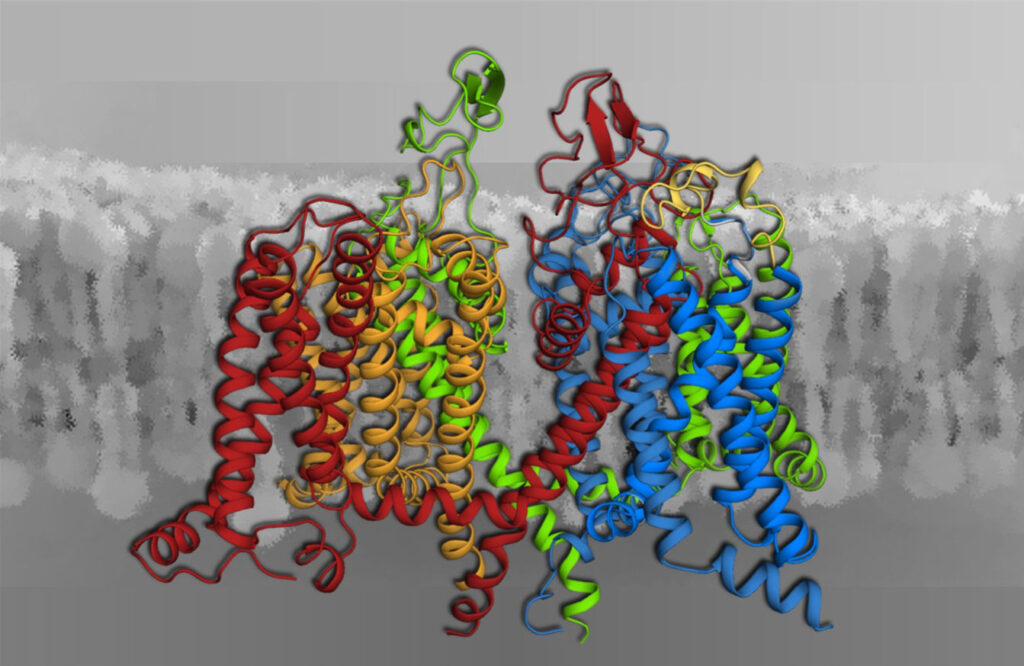
Voltage-gated calcium channels (Fig. 2) are the only proteins capable of sensing electrical signals at the cell surface and convert them into cell functions such as the contraction of skeletal and heart muscle, or the synaptic transmission in the nervous system. Conversely, aberrant calcium channel function is the cause of numerous neurological or psychiatric diseases, and calcium channels are prime targets for therapeutics used to treat such ailments. Our research covers the entire scope from exploring the biophysical mechanisms of channel functions at the molecular level to their roles in human physiology as well as nerve and muscle diseases. We apply state-of-the-art genetic, molecular, biophysical, physiological and computational technologies.
Gastrointestinal Physiology
Univ.-Prof. Dr. Johannes Fürst
The mucosa of the intestines constitutes a selective barrier, which permits nutrient absorption and protection from undesired luminal content. At present, our focus are the function of the non-gastric H+/K+-ATPase ATP12A in regulating the colonic barrier, and the role of selected neuropeptides and cytokines in the physiology of colonocytes. Methods used are transcriptomics, Western blotting, trans-epithelial electrical resistance measurements, histological staining, cell culture and microscopy.
Respiratory Cell Physiology
Univ.-Prof. Dr. Mag. Thomas Haller
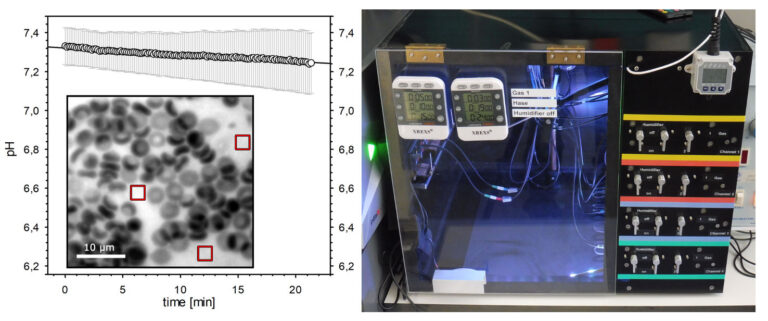
Reversible oxygen binding by haemoglobin is of fundamental importance in medicine and physiology, but has faced technical limitations in the past. We have developed new methods to be used as an analytical service in everyday clinical practice (Fig. 3). Novel aspects of O2 transport in relation to high altitude, accidental hypoxemia, relevant clinical interventions and pharmacological/dietary modulation are explored. Other current studies are devoted to the lifespan of red blood cells and its determinants, using allometric approaches and changes in oxygen affinity during haemodialysis
Human Kidney Model Systems
Univ.-Prof. Dr. Judith Lechner
The research team uses iPSC lines of the IMI – StemBANCC initiative to establish and characterise in vitro model systems for the study of mechanisms of sex differences in the cellular responses to environmental challenges and toxic stimuli. The group promotes the implementation of the SAGER (sex and gender equity in research) guidelines in preclinical research. State of the art information on novel micro-physiological systems and in vitro methodology is offered through student courses and consultation of researchers to replace animal experimentation (https://www.i-med.ac.at/muianimalfree/index.html.de)
Auditory Neuroscience
Dr. Christian Vogl
In the mammalian inner ear, synaptic sound encoding is achieved by ultrafast and tightly regulated neurotransmission between inner hair cell (IHC) ribbon synapses and postsynaptic spiral ganglion neurons. While the mature physiology of these unconventional synapses has been at the focus of current research, fundamental aspects of synaptogenesis and developmental maturation still remain elusive. Therefore, our group employs a comprehensive methodological approach that combines fluorescence live-cell imaging of IHCs – to track developing ribbon synapses in real time – with immuno-histochemical analyses and super-resolution microscopy, to identify the underlying cell biological processes.
Junior Research Groups
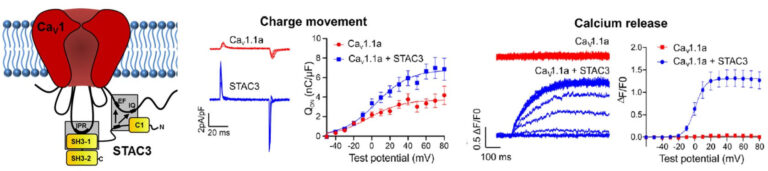
Dr. Marta Campiglio recently identified two distinct molecular interactions between the voltage sensor of excitation-contraction coupling CaV1.1 and the STAC3 scaffold protein. STAC3 has multiple roles in EC coupling, as it is essential for CaV1.1 function as a voltage sensor and for the conformational coupling with the RyR1. Her team is currently investigating the contribution of the two interactions to these multiple STAC3 functions (Fig. 4).
Dr. Theodora Kalpachidou explores the role of microRNAs (miRNAs) in the complex responses to peripheral nerve injury related to neuropathic pain and neuronal regeneration. Several miRNAs that are deregulated after a peripheral nerve injury, and she aims to understand how these small RNA molecules regulate gene networks, how miRNAs controlled mechanisms promote neuro-regeneration, and how they can be developed into novel therapeutic approaches.
Dr. Kai Kummer focuses on plastic changes in the brain that give rise to the psychological comorbidities associated with chronic pain. He explores cholinergic projections from the basal forebrain to the medial prefrontal cortex as a promising pathway in the development of these comorbidities. To gain novel mechanistic insight, he utilises molecular tracing techniques, in vitro electrophysiological recordings, in vivo behavioural assays and opto- and chemo-genetic stimulations.
Pictures
Selected Publications
Zeidler M, Kummer KK, Schöpf CL, Kalpachidou T, Kern G, Cader MZ, Kress M.: NOCICEPTRA: Gene and microRNA Signatures and Their Trajectories Characterizing Human iPSC-Derived Nociceptor Maturation. Adv Sci (Weinh). 2021 Nov;8(21):e2102354. doi: 10.1002/advs.202102354
Kalpachidou T, Malsch P, Qi Y, Mair N, Geley S, Quarta S, Kummer KK, Kress M. Genetic and functional evidence for gp130/IL6ST-induced transient receptor potential ankyrin 1 upregulation in uninjured but not injured neurons in a mouse model of neuropathic pain.
Pain. 2022 Mar 1;163(3):579-589. doi: 10.1097/j.pain.0000000000002402.
May MA, Barré N, Kummer KK, Kress M, Ritsch-Marte M, Jesacher A. Fast holographic scattering compensation for deep tissue biological imaging. Nat Commun. 2021 Jul 15;12(1):4340. doi: 10.1038/s41467-021-24666-9.
El Ghaleb, Y., Schneeberger, P.E., Fernández-Quintero, M.L., Geisler, S.M., Pelizzari, S., Polstra, A.M., van Hagen, J.M., Denecke, J., Campiglio, M., Liedl, K.L., Stevens, C.A., Person, R.E., Rentas, S., Marsh, E.D., Conlin, L.K., Tuluc, P., Kutsche, K., and Flucher, B.E. (2021) CACNA1I gain-of-function mutations differentially affect channel gating and cause neurodevelopmental disorders. Brain, 144:2092-2106. doi: 10.1093/brain/awab101.
Fernández-Quintero, M., El Ghaleb, Y., Tuluc, P., Campiglio, M., Liedl, K.R., and Flucher, B.E. (2021) Structural determinants of voltage-gating properties in calcium channels. eLife, doi: 10.7554/eLife.64087.
Kaplan, M.M. and Flucher, B.E. (2022) Counteractive and cooperative actions of muscle β -catenin and CaV1.1 during early neuromuscular synapse formation. iScience, 25:104025. doi: 10.1016/j.isci.2022.104025.
Chandrasekaran, V; Carta, G; da Costa Pereira, D; Gupta, R; Murphy, C; Feifel, E; Kern, G;
Lechner, J; Cavallo, A-L; Gupta, S; Caiment, F; Kleinjans, J; Gstraunthaler, G; Jennings, P; Wilmes, A Generation and characterization of iPSC-derived renal proximal tubule-like cells with extended stability. Sci Rep 11, 11575 (2021). https://doi.org/10.1038/s41598-021-89550-4
Woyke S, Ströhle M, Brugger H, Strapazzon G, Gatterer H, Mair N, Haller T.: High‐throughput determination of oxygen dissociation curves in a microplate reader—A novel, quantitative approach Physiological Reports 2021;9:e14995
Selection of Funding
Stand-alone projects:
- FWF, P30402: Regulation of voltage-sensitivity of CaV calcium channels (Flucher)
- FWF, P35618: Regulation of Ca currents and EC coupling by CaV1.1 voltage sensors (Flucher)
- FWF P33270: The role of CaV1.1 calcium channels in presynaptic differentiation during neuromuscular junction development (Flucher)
- FWF, P33776, Analysis of the functions of multiple STAC interactions (Campiglio)
- FWF, P30809: DynChomiR (Kummer)
- FWF, P34403: POCDMiBiOME (Kress)
- DFG, VO 2466/1-1, Paralemmin-3 – ein essentieller Baustein des submembranösen Zytoskeletts in auditorischen Haarzellen (Vogl)
- FWF, P36229: GAPmir: A novel miRNA in the GAP43 gene regulating neuroregeneration (Kalpachidou)
DocFunds:
- FWF, DOC-30: PhD program: Calcium channels in excitable cells (Speaker: Obermair, PIs: Campiglio, Flucher) until 2022
- FWF, DOC-178: PhD program: Calcium channels in excitable cells (PIs: Campiglio, Vogl)
Consortia:
- European Commission, MSCA-ITN “TOBeATPAIN” GA764860 (Kress, PI)
- FWF, I050870: PainMSK (Kress, PI)
- FWF, I5364: LIAISON: Neuartige Interaktion von Kv Kanälen (Kummer, PI)
Translational projects:
- AWS Prototypenförderung (T. Haller, S. Woyke)
Collaborations
- Hermann Brugger, Eurac Research, Bozen, Italy
- Klaus Liedl, University of Innsbruck, Austria
- Marzia Malcangio, King’s College London, United Kingdom
- Markus Missler, Westfälische Wilhelms-University, Münster, Germany
- Tobias Moser, Institute for Auditory Neuroscience, University Medical Center Göttingen, Germany
- Theodore J. Price, University Dallas, USA
- Markus Ritter, Paracelsus Medical University, Salzburg, Austria
- Hermona Soreq, Hebrew University Jerusalem, Israel
- Jörg Striessnig, University of Innsbruck, Austria
- Filip Van Petegem, University of British Columbia, Canada
- Hermann Brugger, Eurac Research, Bozen, Italy
- Peter Kotanko, Renal Research Institute, New York, USA
- André Stadler, Alpenzoo Innsbruck, Austria